
This is the accurate way of rounding off to the correct number of significant figures in multistep calculations. I do it here mainly to save space, but you can keep the numbers in the calculator and round them off at the end. 
The little inconsistency in the final answer is due to rounding off the intermediate numbers. So, to calculate the moles from a given mass, we make a conversion factor correlating 1 mole with the molar of the given component.įor example, how many moles of sulfur are there in a 16.2 g sample? The molar mass, on the other hand, is a constant number for a given atom or a molecule as it is for a specific amount of it. The given mass is the mass of the sample, and it can be any number, for example, we can have 10 g of salt, 15 g, or 100 g. To calculate the moles from a given mass (m), the molar mass of the component is used. The molecular mass of water is 18.0 amu, and the molar mass is 18.0 g/mol.įor the molar mass, keeping one decimal is usually an acceptable approximation, and for the Avogadro’s number, you can use 6.02 x 10 23. Numerically, the molar mass is equal to the atomic mass of a given atom or a molecule, so we can look up the molar mass of an element in the periodic table.įor example, we can say that the mass of Cu is 63.55 amu or 63.55 g/mol. The mass of one mole of atoms/molecules/ions is called its molar mass (M) expressed in g/mol. One mole of any element contains 6.022 x 10 23 of its atoms, and a mole of a compound contains 6.022 x 10 23 molecules, ions-ions, and in general, 1 mol = 6.022 x 10 23 particles. This is called Avogadro’s number ( N A = 6.022 x 10 23). By definition, a mole is the number of atoms in a 12-g sample of 12C isotope which happens to be 6.022 x 10 23 atoms. R Range in isotopic composition of normal terrestrial material prevents a more precise Ar(E) being given the tabulated Ar(E) value and uncertainty should be applicable to normal material.The mole is just a number like a dozen, a hundred, or a million. Substantial deviations in the atomic mass of the element from that given in the table can occur. M Modified isotopic compositions may be found in commercially available material because it has been subjected to an undisclosed or inadvertent isotopic fractionation. The difference between the atomic mass of the element in such specimens and that given in the table may exceed the stated uncertainty. G Geological specimens are known in which the element has an isotopic composition outside the limits for normal material. †Commercially available Li materials have atomic weights that range between 6.939 and 6.996 if a more accurate value is required, it must be determined for the specific material. However, three such elements (Th, Pa, and U) have a characteristic terrestrial isotopic composition, and for these an atomic mass is tabulated. 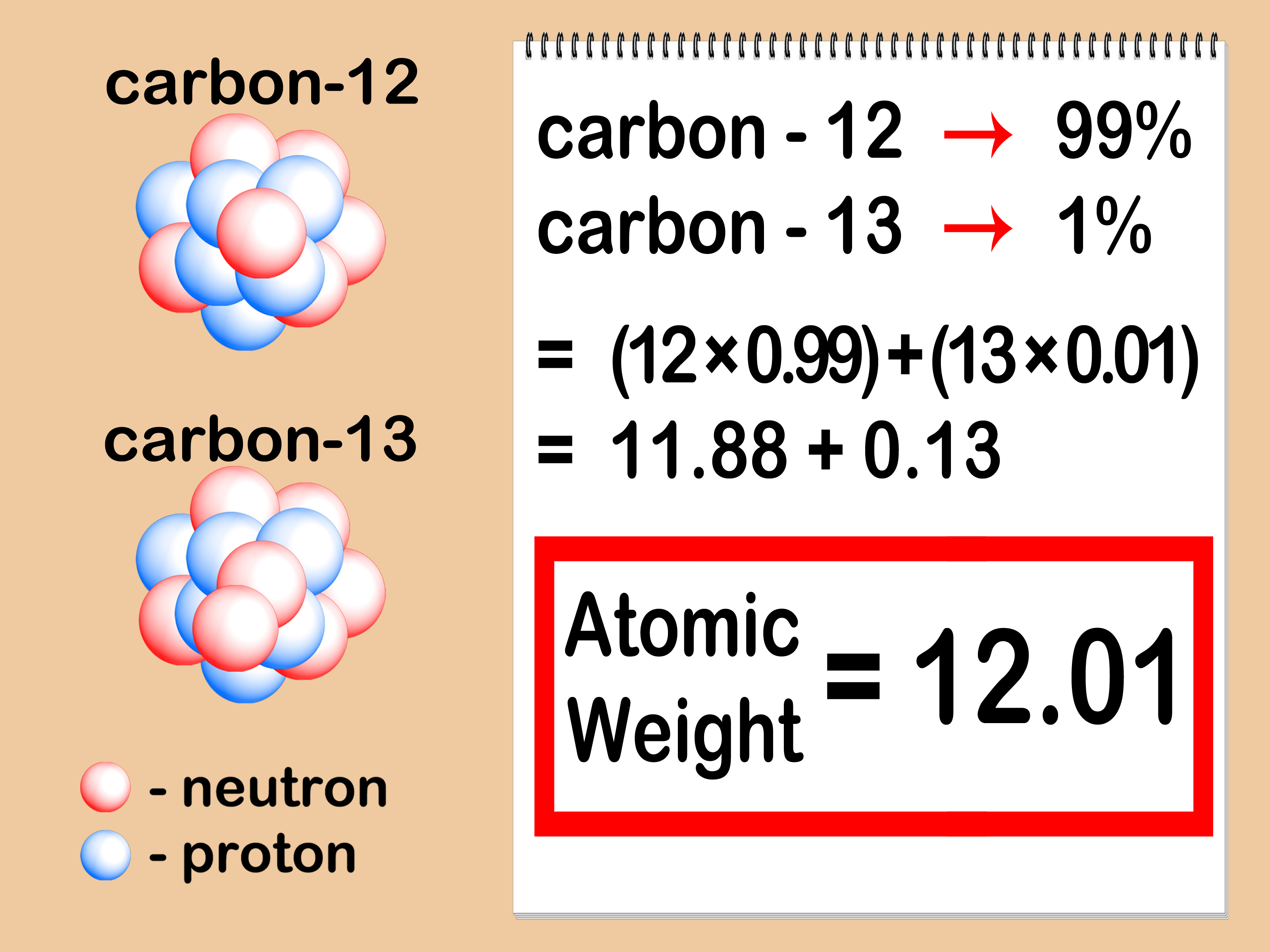
Table 21.1 The Basics of the Elements of the Periodic Table Name The web offers many interactive periodic table resources. Some elements, such as lithium, can have varying atomic masses depending on how their isotopes are isolated. The atomic masses of other elements, especially radioactive elements, are not known as precisely. The atomic masses of some elements are known very precisely, to a large number of decimal places. The atomic masses in Table 21.1 "The Basics of the Elements of the Periodic Table" represent the number of decimal places recognized by the International Union of Pure and Applied Chemistry, the worldwide body that develops standards for chemistry. However, some applications (especially nuclear chemistry see Chapter 11 "Nuclear Chemistry") require more decimal places. Or two decimal places are necessary for the atomic mass. One item on most periodic tables is the atomic mass of each element. The Internet is a great place to find periodic tables that contain additional information. What follows here is a relatively simple version. Periodic tables are available with a variety of chemical and physical properties listed in each element’s box. Many of the physical and chemical properties of the elements are either known or understood based on their positions on the periodic table. 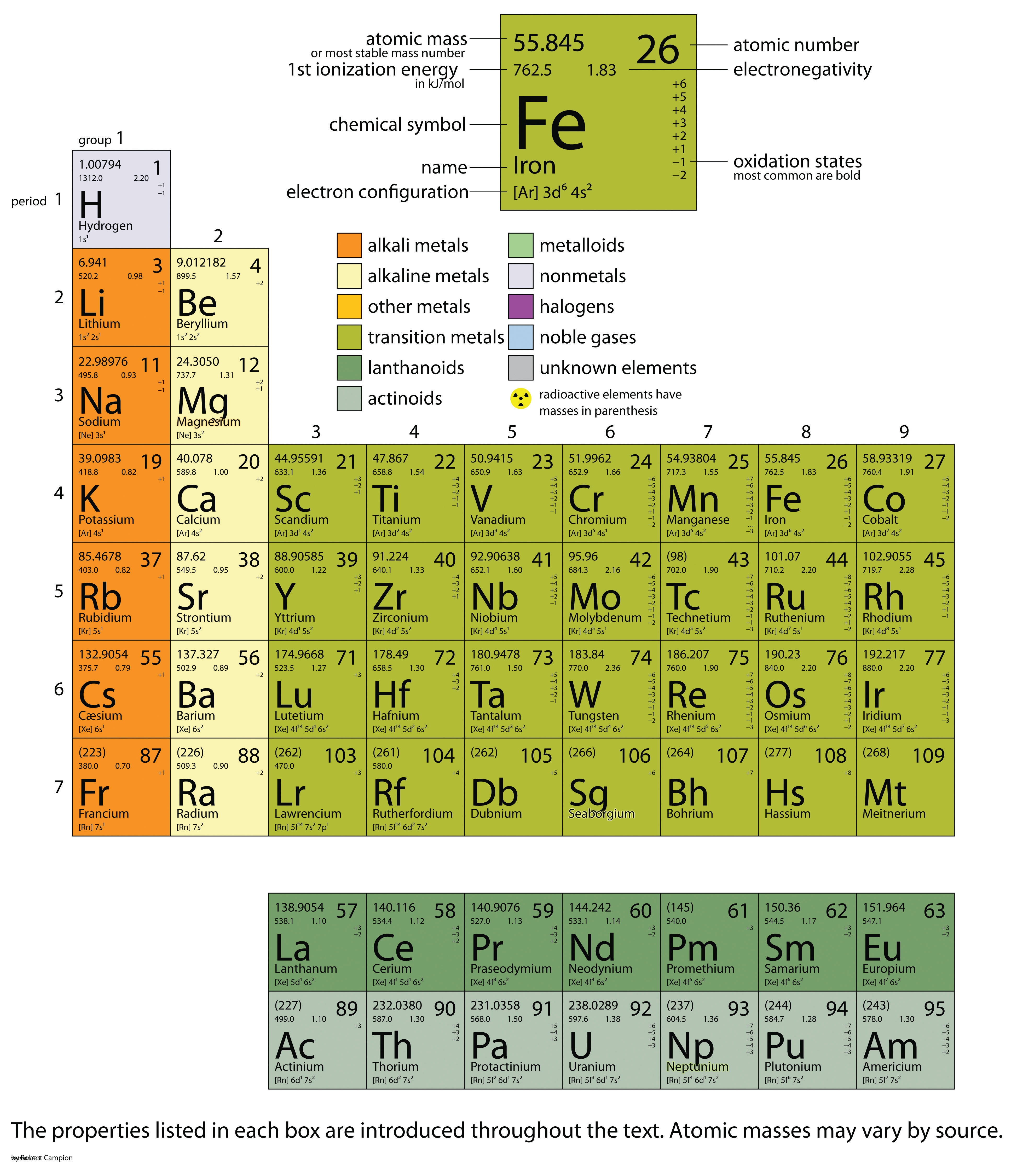
The periodic table is arguably the best tool in all of science no other branch of science can summarize its fundamental constituents in such a concise and useful way. The periodic table listsĬhemical elements, arranged by atomic number (that is, the number of protons in the nucleus). In this chapter, we present some data on the chemical elements. Chapter 21 Appendix: Periodic Table of the Elements
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
